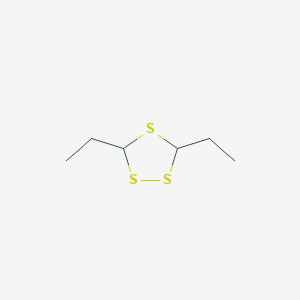
3,5-Diethyl-1,2,4-trithiolane
| Common Name: |
3,5-Diethyl-1,2,4-trithiolane |
| IUPAC Name: |
3,5-diethyl-1,2,4-trithiolane |
| Molecular Formula: |
C7H14O3 |
| SMILES: |
CCC1SC(SS1)CC |
| Inchi: |
1S/C6H12S3/c1-3-5-7-6(4-2)9-8-5/h5-6H,3-4H2,1-2H3 |
| Inchi Key: |
WQXXXHMEBYGSBG-UHFFFAOYSA-N |
| Cas No: |
54644-28-9 |
| Name |
Value |
| Lipinski Violations |
0 |
| Ghose Violations |
2 |
| Veber Violations |
0 |
| Egan Violations |
0 |
| Muegge Violations |
1 |
| Name |
Value |
| Molecular Weight (g/mol) |
146.18 |
| Mass (g/mol) |
180.01 |
| Molar Refractivity |
38.21 |
| Net Charge |
|
| HBD |
1 |
| HBA |
3 |
| Rt Bonds |
4 |
| Rings |
1 |
| TPSA |
46.53 |
| Hetero Atoms |
3 |
| Heavy Atoms |
10 |
| Aromatic Heavy Atoms |
0 |
| Melting Point (°C) |
|
| Boiling Point (°C@760.00mm Hg) |
70.00 to 72.00 @ 1.00 mm Hg |
| Vapor Pressure (mmHg@25.00 °C) |
0.018 |
| Vapor Density (Air =1) |
|
| Fraction Csp3 |
0.86 |
| LogP |
3.587 |
| iLOGP |
2.03 |
| XLOGP3 |
1.19 |
| WLOGP |
0.57 |
| MLOGP |
0.75 |
| ESOL Log S |
-1.23 |
| ESOL Solubility (mg/ml) |
8.57 |
| ESOL Solubility (mol/l) |
0.059 |
| ESOL Class: esol_class |
Very soluble |
| Ali Log S |
-1.76 |
| Ali Solubility (mg/ml) |
2.52 |
| Ali Solubility (mol/l) |
0.02 |
| Ali Class |
Very soluble |
| Silicos-IT LogSw |
-0.64 |
| Silicos-IT Solubility (mg/ml) |
33.8 |
| Silicos-IT Solubility (mol/l) |
0.23 |
| Silicos-IT Class |
Soluble |
| Name |
Value |
| GI Absorption |
High |
| BBB Permeable |
1 |
| PgP Substrate |
0 |
| Log Kp (cm/s) |
-6.35 |
| Bioavailability Score |
0.55 |
| Caco2 |
1 |
| Human Intestinal Absorption |
1 |
| Plasm Protein Binding |
0.643 |
| CYP1A2 Inhibitor |
0 |
| CYP2C19 Inhibitor |
0 |
| CYP2C9 Inhibitor |
0 |
| CYP2D6 inhibitor |
0 |
| CYP3A4 inhibitor |
0 |
| Ames mutagenesis |
0 |
| Acute Oral Toxicity |
2.789 |
| Carcinogenicity (Binary) |
0 |
| Carcinogenicity (Trinary) |
Non-required |
| Eye Irritation |
1 |
| Hepatotoxicity |
0 |
| Androgen Receptor Binding |
0 |
| Aromatase Binding |
0 |
| Estrogen Receptor Binding |
0 |
| Glucocorticoid Receptor Binding |
0 |
| Thyroid Receptor Binding |
0 |
| BRCP inhibitor |
0 |
| BSEP inhibitor |
0 |
| OATP1B1 inhibitor |
1 |
| OATP1B3 inhibitor |
1 |
| OATP2B1 inhibitor |
0 |
| OCT1 inhibitor |
0 |
| OCT2 inhibitor |
0 |